 Interactive periodic table with up-to-date element property data collected from authoritative sources. As chlorine can easily accept an electron as compared to phosphorus and sulphur, the chemical reactivity increases from phosphorus to chlorine. Please enable Javascript in order to use PubChem website. The activity series is a list of elements in decreasing order of their reactivity. Chlorine (Cl) has 7 valence electrons and needs only one more electron to complete its octet. Sulphur (S) has 6 valence electrons and needs 2 more electrons to complete its octet. Phosphorus (P) has 5 valence electrons and needs 3 electrons to complete its octet. Moving further right in the period towards non-metals, the chemical reactivity again gradually increases. This is because the number of valence electrons increases, making it difficult to lose electrons. The general reaction for alkali metal hydrides and water is given below: (4) M H ( s) + H 2 O ( l) M ( a q) + + O H ( a q) + H 2 ( g) This reaction can be. In this case, however, hydrogen gas is produced with the metal hydroxide. This could be used to follow up some work on the periodic table where the trends in reactivity in groups 1 and 7 have been identified. Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons. Similarly to the Group 1 oxides, the hydrides of the Group 1 elements react with water to form a basic solution. Chemistry for the gifted and talented: trends in reactivity in the periodic table. A chemical that reacts easily with other substances is considered to be highly reactive. Explain they will be looking at trends on the periodic table, one of which is the reactivity of elements. Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons. On the board, list everything students already know about the periodic table. But, at the far right are the noble gasses, and they are almost completely inert. are found on the left and in the middle of the periodic table. Elements cannot be broken down into simpler substances. Each element going from left to right tends to be more reactive. Elements are listed on the periodic table. The reactivity of metals increases further left along a period, and further down a group. Reactivity: The reactivity of the elements increases going from left to right on the periodic table. The chemical reactivity gradually decreases as we go to aluminium (Al) and silicon (Si). Another common method of categorization recognizes nine element families: Alkali Metals: Group 1 (IA) - 1 valence electron. Reactivity is dependent on the classification of an element (metals and non-metals), as they both have differing periodic trends. 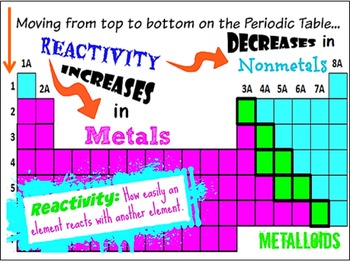
On moving from left to right in a period of the periodic table, the chemical reactivity of the elements first decreases and then increases.įor example, in the third period elements comprising of Na, Mg, Al, Si, P, S and Cl, sodium (Na) is very reactive as it has one valence electron, and can therefore, lose its electron easily. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
